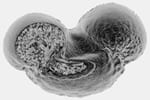
Here is an autofluorescent pollen grain, from the Carolina Biologicals’ “mixed pollen” specimen. Click on the little pictures to download quicktime animations of the section series (1 meg) and the surface-rendering (2.5 megs). Images were processed using Molecular Dynamics ImageSpace software.
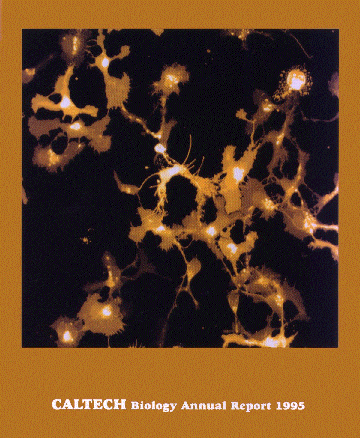
Dissociated Neural Cell Culture
Front cover of the Biology Division Annual Report. Living rat hippocampal neurons were stained with DiO, and imaged using pulsed illumination at 900 nm. These were imaged one day after staining and plating onto a poly-lysine-coated plastic Petri dish. The image is a projection through 50 sections of 0.3 um each. No dye bleaching was observed during scanning. The imaging had no adverse effect on the health of the cells, compared to unscanned regions in the same dish after another day in culture.
Living rat hippocampal neurons sending processes out from wells of a Neuroprobe. The upper photograph was taken using Nomarski optics one day after staining neurons with DiI and placing them into the probe wells, and shows larger neurites growing across the silicon substrate. The lower image is a projection through 10 1-um slices scanned by the 2-photon scope at 900 nm excitation. Fine processes and growth cones are visible, as are the cell bodies trapped beneath the transparent silicon nitride grillwork. The neurons were still growing happily the next day, unaffected by 2-photon imaging.
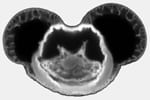
Comparison of Confocal and Two-Photon imaging. A single pollen grain was imaged using visible excitation in confocal mode (CLSM) or using pulsed IR excitation in 2-photon mode (TPLSM). The pollen is a sphere with spikes pointing in all directions. The top two images are single horizontal slices through the base of the uppermost spikes. One can see more out-of-focus spikes in the CLSM image than in the TPLSM image, despite the fact that the smallest (50 um) pinhole was used for the left image, and no aperture (6 mm “open” setting) was used for the right image. The lower two images show vertical cross-sections through stacks of horizontal slices through the thickness of the pollen grain. Not only is the TPLSM image better resolved than the CLSM image, but the enhanced penetration of the IR excitation beam is also evident; spikes are visible on the underside of the grain in the TPLSM but not in the CLSM image. Scale bar, 20 um.
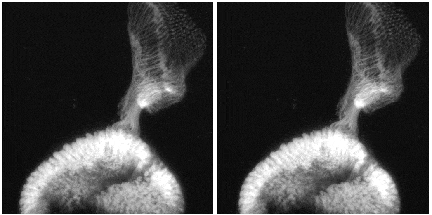
Stereo pair of living fruit fly optical projections labeled with Green Fluorescent Protein by Paul Garrity at the UCLA Howard Hughes Medical Institute, imaged at Caltech using 900 nm pulsed excitation. The optic disk of the eye, at the bottom of the image is approximately 200 um across. Individual growth cones of the photoreceptor neurons are clearly visible as they project to their target in the medulla, at the top of the image.
- Return to the 2-Photon pages
- To Steve Potter’s home page